
 |
Air conditioning - 2. The self contained system |
I will describe two typical systems. The first is air conditioning for a single room, the second is a full system for a large building. Most of the available literature, particularly on the internet, takes it for granted that air conditioning is a good thing and then proceeds to tell you how to design it. This article is designed to help you understand other people's designs, and judge their suitability for your purpose.
A fan draws air from the room first through a cooling device, consisting of metal fins extending from a pipe through which cooling fluid circulates, at a rate determined by the thermostat or by the humidistat. The air next passes over a heater, usually electrical, which is energised on instructions from the room thermostat.
Air conditioning has in the past been used where the climate is too hot for comfort. Cooling will increase the relative humidity of the air, so humidification is not usually built into these systems. If it is necessary, the usual method is to inject steam from electrically boiled water.
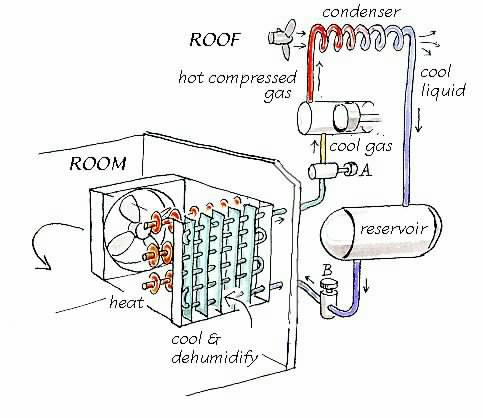
Figure 2. Minimal air conditioning. The part of the system in the room, on the left, pulls air first over a cool surface and then over a warming surface. The part of the system on the right recirculates the cooling fluid. The fluid passes from the reservoir through a valve B into the lower pressure within the cooling unit in the room. There the liquid boils, removing heat from the air. The boiling point is fixed by the constant pressure set by valve A. The vapour is then compressed and condensed back into a liquid which collects in the reservoir ready for another cycle.
That is all there is to the part of the system in the room, which is sketched on the left in figure 2. The bit that is more difficult to understand, or at least unfamiliar to most people, is how the cooling fluid is produced and controlled. That is the part on the right of the diagram.
The cooling fluid used to be a chlorofluorocarbon compound, and often still is, though they all more or less ravage the earth's ozone layer. The essential characteristics of these fluids is that they have quite a low boiling point at atmospheric pressure and that they can stay in the pipes for a long time without decomposing either themselves or the pipes. Finally they need to have some lubricating ability, or the ability to carry a lubricant, because the fluid has to be compressed and pumped round the system. This rare set of necessary properties has proved difficult to combine with friendliness to the earth's atmosphere.
The liquid is let into the cooling unit through a valve marked B on the diagram. It evaporates while it passes through the pipe, taking heat from the air just as water evaporating from a towel laid on your fevered brow cools you when on holiday in the Mediterranean. The temperature in the cooling coil depends partly on the amount of fluid let in by the valve, which is controlled by the thermostat or the humidistat. But now comes a crucial difference from your Mediterranean experience: the minimum temperature at the cold surface can be fixed by controlling the pressure in the cooling coil, with the valve marked A on the diagram. The boiling point of any liquid depends on the pressure. One could use water in the cooling coil, if the pressure is kept low enough. At 1000 Pa pressure, which seems a lot but is just 1% of atmospheric pressure, water boils at 7 degrees. It isn't used in cooling coils of this evaporative type because it has practical disadvantages.
The reason for wanting to limit the minimum temperature is to stop ice clogging the air passage. There are clever systems which notice when ice has formed and hold a melting pause, but that adds to the cost. The pressure controller is therefore set to make the cooling fluid boil at the lowest temperature that is likely to be needed to control the humidity, but always over zero degrees. The temperature needed for cooling is nearly always higher than that needed for dehumidification so it is the RH setting that is decisive.
This brings me to the first point that conservators need to understand: it is expensive to produce air at a dew point below about 4 degrees in this type of equipment. This dewpoint corresponds to 50% RH at 15°C. This sort of air conditioning is entirely suitable for keeping people comfortable but it is not good for specialised stores, for films or for furs, for example, where one needs a temperature below 15 degrees. Such equipment is, however, often used for such places. A better solution is to use an absorption dehumidifier, which will be described in a later article.
Now back to the main story: The vapour that emerges through the pressure controller is gathered up by a compressor. The compression also heats the gas, as will be understood by anyone who has pumped up a cycle tyre. The hot gas is then led away from the room, to be cooled down. This is often done on the roof or in a small enclosure which vibrates to the roar of the fan blowing air over the fins of a condenser. The cooled, now liquid coolant is piped back to the reservoir, ready for its next tour through the room air conditioner.
The entire process described above is inefficient and uses electricity, which is itself produced by inefficient conversion of heat energy. Such systems are therefore confined to small places where the inefficiency is compensated by the generally high reliability and freedom from maintenance.

This work is licensed under a Creative Commons Attribution-Noncommercial-No Derivative Works 3.0 License.