
 |
The response to changing temperature of a back-protected canvas |
Tim Padfield, Nicolas Padfield,
Anne Thøgersen, Astrid Valbjørn Nielsen,
Cecil Krarup Andersen and Mikkel Scharff
October 2019
Complete data to support this article (zip).
A painting on canvas, stretched on a wooden frame, was fitted with various styles of back protection and then exposed to a cycle of temperature variation at the back, with the front exposed to a constant room temperature. The painting was also exposed with a constant wall temperature and varying room temperature. The space between the canvas and the back board was fitted with temperature and relative humidity (RH) sensors.
The unprotected painting suffered a large RH variation, because of the varying canvas temperature interacting with the constant room air moisture content.
Effective stabilisation of the RH behind the canvas against temperature variation is provided by a shiny aluminium alloy sheet sealed against the frame.
Convective mixing of the air within the enclosed space results in the inside surface of the non-absorbent back board experiencing a strong variation in RH, because of buffering of the space by the painting canvas at a different temperature.
Either a space or insulation between this back plate and the wall will reduce the risk of condensation on the inner surface of the back plate. It will however increase the risk of condensation on the wall surface behind the painting.
An absorbent back board stabilises its own surface RH but de-stabilises the RH at the painting canvas surface. It is unwise to use a moisture-active back board where there is likely to be a temperature gradient between a painting and its back board.
If protection is needed against a hostile room RH, extra humidity buffer can be placed behind the painting canvas but it must be kept close to the painting temperature by insulation between buffer and back board. This increases the temperature and the RH variation at the back board and thus increases the risk of condensation on its inner surface.
The RH and the temperature in the narrow space between the canvas and the wooden stretcher are always more nearly constant than in the open canvas area, which suggests an explanation for the widely observed better condition of the areas of canvas paintings which lie close over the support structure.
Paintings set against uninsulated outer walls are exposed to a varying temperature gradient. Absorbent and insulating materials within the space enclosed behind the painting affect the moisture content of the painting support in quite a complicated way. This phenomenon is explored through a series of experiments with various arrangements of back protection.
The emphasis is on understanding the micro-environment, using materials and environmental variations which simplify the interpretation. The practical implementation of the physical principles is discussed at the end of the article.
Temperature variation in spaces with a uniform water vapour concentration, which is the normal situation, is a potent cause of mould growth and humidity induced damage. This is because the surface relative humidity (RH) at a relatively cold surface can rise very high in spaces which contain a moisture reactive material at a higher temperature.
There is abundant advice about back protection of paintings. However, it neglects the disturbing influence of a temperature gradient on the RH near the painting and gives the impression that adding buffer material at the backboard or within the enclosure is always beneficial.
Sozzani (Sozzani 1997) summarises the effect of temperature change on the RH within a vitrine enclosing a painting. He states:
“Preliminary testing of various backing board materials indicates that the more hygroscopic the backing board the greater the overall buffering effect."
The Canadian Conservation Institute provides advice on back board protection of paintings (Hartin 2016) which includes this passage:
“The board can also be used in combination with a hygroscopic material capable of absorbing and releasing moisture.” And further: “Tests have indicated that synthetic boards combined with a hygroscopic material, such as a sheet of 4-ply matboard, will provide additional control of the RH within the space that is enclosed by the painting and the backing board. This hygroscopic layer will release moisture as required in response to environmental conditions, thereby helping prevent the low RH conditions that are detrimental to the painting’s paint, ground and size layers.”
Sandner et al. (Sandner, Schramn, and Schaft 1993) advocate a back protection of polyurethane foam sheet, with a layer of Artsorb behind the painting as humidity buffer.
Portsteffen (Portsteffen 2016) writes “Hygroscopic backing boards can slow down the hygric changes from the back and will act ideally in the same damping effect as the paint layer. When using hygroscopic cardboards it is wise to build in a distance, as those materials can work like a sponge and behave as a moisture source. To avoid this effect, the use of wooden backing boards is advisable. The wooden backing will slowly transmit the humidity and will act with a true buffer effect serving as a protective damping barrier."
Michalski (Michalski 2005) in his risk analysis of backing boards for paintings, seems not to have considered a temperature gradient between canvas and the wall behind it as a risk.
The literature, and the advice, on humidity stabilisation of works of art concentrates on the use of hygroscopic materials to compensate for slow exchange of moisture with an ambient atmosphere of unsuitable RH, with implicit expectation that the temperature will remain constant, or at least uniform, over the immediate environment. There are scarcely any discussions of the effect on the canvas of a varying temperature or a temperature gradient between painting and wall.
Ligterink and Di Pietro (Ligterink and Di Pietro 2007) examined the influence of temperature fluctuations on the ‘stretcher effect’, which is the better preservation of paintings where the canvas is over the wooden support structure. Their temperature gradient was imposed as a constant difference. Their argument is that the wooden frame provides thermal insulation, thus diminishing the change in RH at the nearby canvas surface by reducing temperature variation. They claim that moisture exchange between wood, canvas and air does not play a significant part in the process.
Padfield et al (T. Padfield et al. 2002) measured moisture movement in glazed prints set against outer walls of houses. These in situ measurements show great variation in RH within the painting enclosure as a consequence of temperature gradients caused by cold walls, and by sunlight on the image. The data were unsuited to interpretation in detail, given the variability of the natural environment.
In this article we aim to fill a hole in the conservation literature by exploring the climate within an enclosed painting where moisture leakage to the surroundings is slow, so changes to the canvas surface RH and its moisture content are due to temperature change alone. The experiments were made in a strictly controlled environment, to approximate reality, but with imposed simplicity to identify subtle processes usually masked by the variation of the weather.
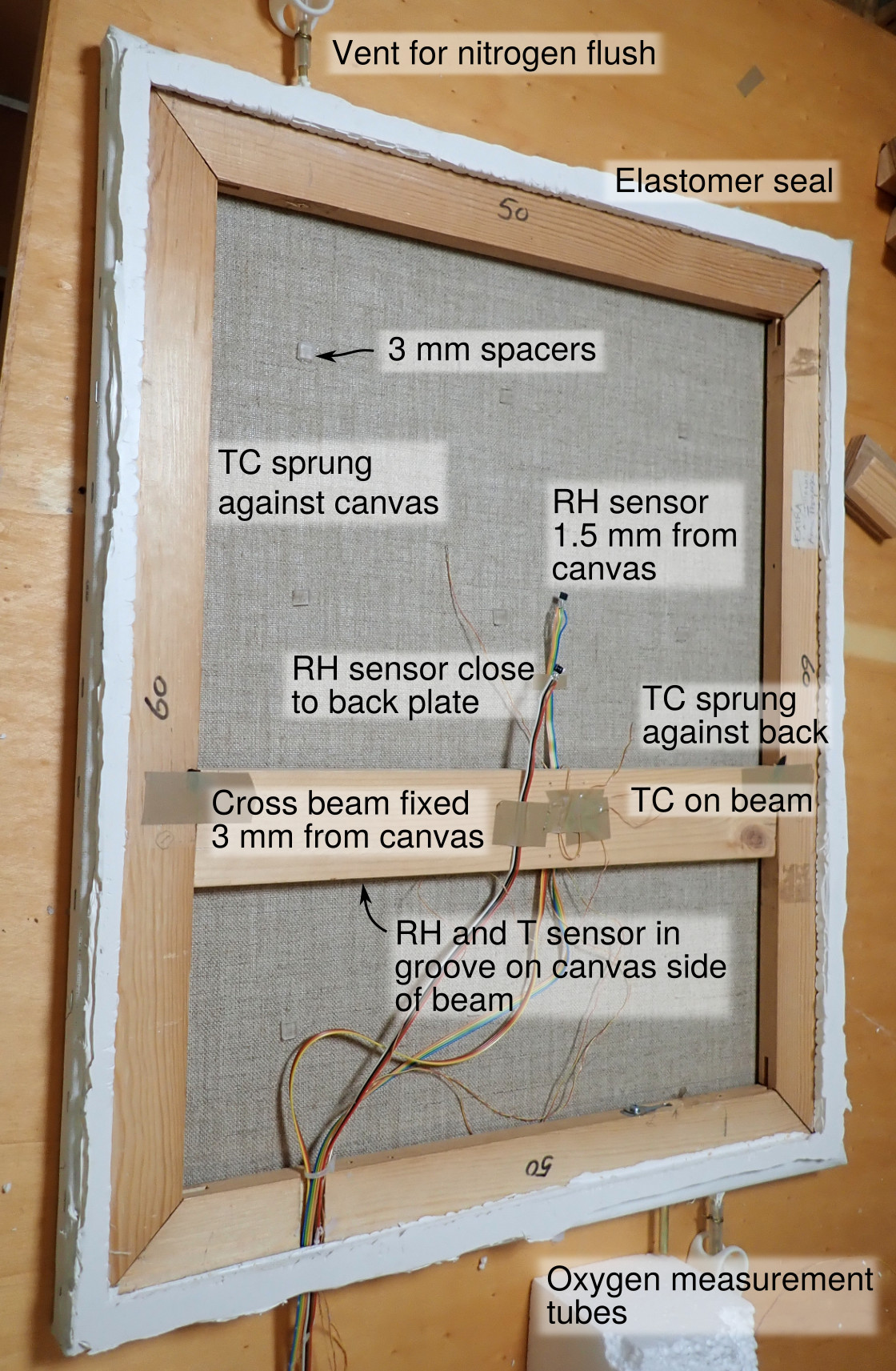
Figure 1. A back view of the experimental canvas stretched over a wooden frame. The fine thermocouples (TC) which spring against the painting canvas and the back plate are scarcely visible. The RH sensors have built-in temperature measurement. There is a third RH sensor mounted in a shallow groove in the cross beam, facing the canvas. The painting dimensions are 600 × 500 mm, the total thickness of the assembly, including an aluminium back plate, is 30 mm.
The canvas was stretched over a conventional softwood frame. Staples were used to hold the canvas to the frame. A coat of white acrylic paint was applied to the outside surface. Its uniformity of colour prevented temperature gradients in the plane of the canvas. An airtight seat for the back plate was essential. To make this, a temporary frame of 6 mm thick wood was prepared, 10 mm smaller than the main frame at each edge. It was covered with polyethylene film and centered over the back of the frame.
A polyurethane type of flexible sealant was squeezed into the corner formed by the canvas edge and the temporary frame. While the sealant was still soft, a polyethylene covered glass plate was gently pressed down over the secondary frame to give an absolutely flat surface to the sealant.
After two days the glass was removed and the polyethylene peeled back to reveal a perimeter gasket which would give an airtight but non-adhesive connection to the aluminium back plate which was used for most of the experiments.
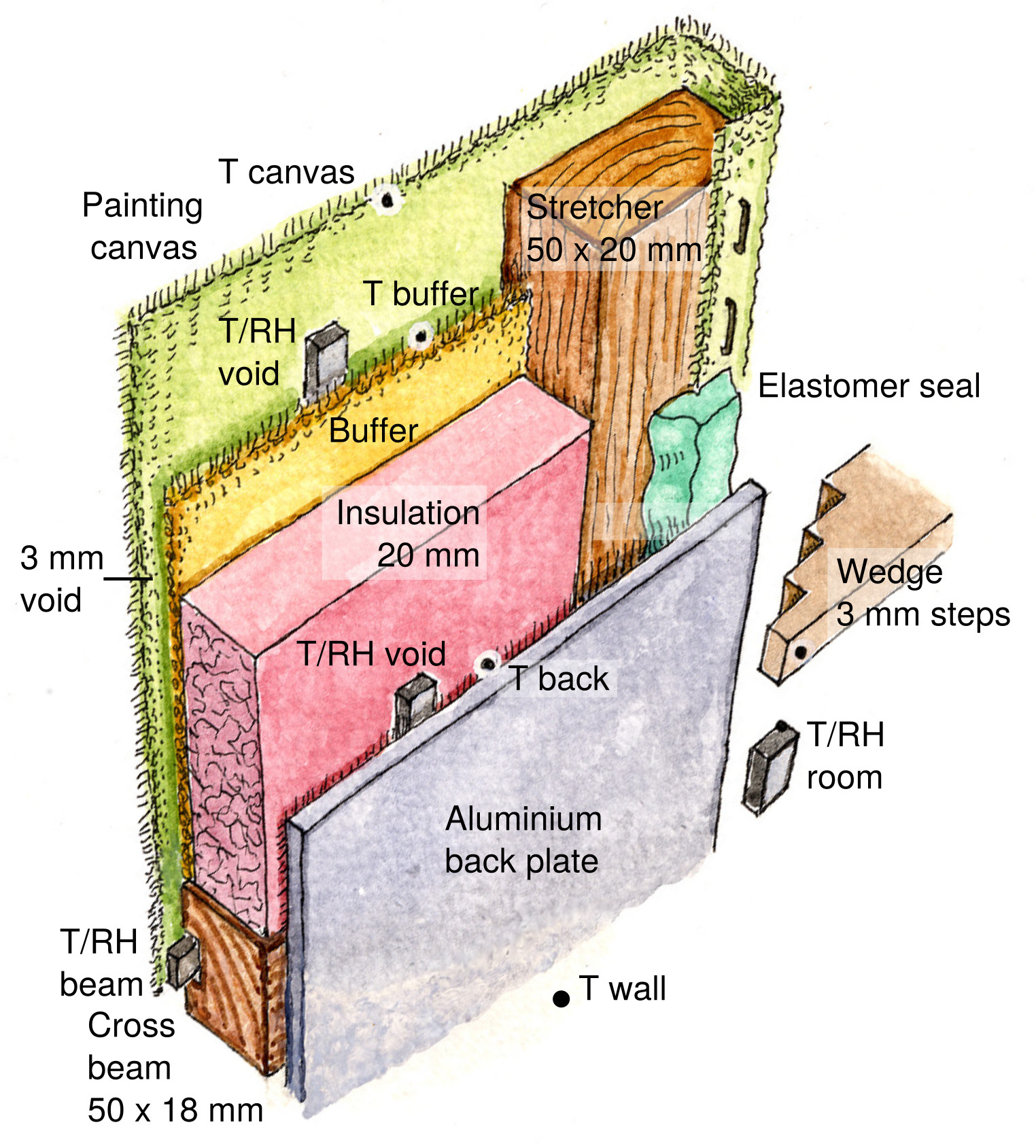
Figure 2. Positions of the components and sensors within and around the painting, viewed from the wall side. The stepped wedge, one of four, is to hold the assembly at a specified distance from the wall.

Figure 3. The experimental painting mounted vertically against a “wall” of painted aluminium which is enclosed flush with a plywood frame.
The painting is held against a vertical painted aluminium plate whose temperature can be varied between 12 °C and 40 °C. On the right in figure 3 is the apparatus for recirculating tempered water through a labyrinth of copper piping behind the plate. The datalogger is near the bottom right corner of the plate.
The painting was exposed to a sinusoidally varying temperature of the “wall” at its back, with a nearly constant room temperature at its front. Several cycles were measured for each frame construction, some of a four hour period, to detect rapid effects, some of 24 hours to imitate a real situation. Two typical cycles were extracted for graphical display. In some experiments the painting was set to face the aluminium plate while the back plate was fanned with room air, to simulate a varying room temperature against a constant wall temperature.
The relative humidity is the quantity of most significance in this article. It is the fundamental expression for the potential for water vapour to enter into chemical reactions, cause dimensional change and facilitate biological processes. It is identical to the chemists’ concept of water activity, which is defined as the ratio of the actual partial pressure of water vapour in space to the partial vapour pressure over a pure water surface at the same temperature. From this value one can derive quantitatively the change of mechanical properties which cause deterioration of paint layers on canvas, and the risk of fungal growth on the canvas and other parts of the painting assembly.
Within the space between the painting and the back plate, the concentration of water molecules by weight, expressed as a ratio to the weight of dry air, is the quantity which tends towards a uniform value over the entire space, even in a temperature gradient. This is called the mixing ratio (MR) and is displayed on the lower portion of the graphs.
To interpret the processes at work one has to switch back and forth between MR and RH. The first to describe the migration of water molecules in space, the second to describe migration of water molecules through the solid to gas interface.
It is difficult to be sure of the RH at a surface, particularly if that surface is moisture reactive.
The boundary layer of still air ‘glued’ to a surface is quite thin, typically less than 3 mm, even in the apparently still air in an enclosure. The temperature gradient, and the vapour concentration gradient, across this boundary layer is steeper than in the rest of the enclosure, because this thin layer is a good insulator compared with the convecting flow of air further away.
Even miniature RH sensors are bulky, 5 × 4 × 2 mm, and obstruct the movement of heat and moisture at the point where they measure.
In these experiments we partially compensate for this practical difficulty by measuring both temperature and relative humidity at a point about 1.5 mm away from the surface, while also measuring the surface temperature nearby with a thin thermocouple, which hardly interferes with the flux of moisture and heat at the surface.
We then estimate the expected RH at the surface by dividing the mixing ratio of the air at the RH sensor by the mixing ratio of vapour-saturated air at the surface temperature.
This calculation assumes that there is no significant vapour flux through the surface into the boundary layer. In the experiments described below, this is only true for the aluminium surface of the backing plate. At the canvas surface our RH extrapolation will be wrong, but better than quoting the RH at a distance.
The magnitude of the difference between the surface RH and the RH at a millimetre or two from the surface is significant in interpreting the experimental results. In the graphs, the calculated surface value and the measured value at the RH sensor are linked by shading.
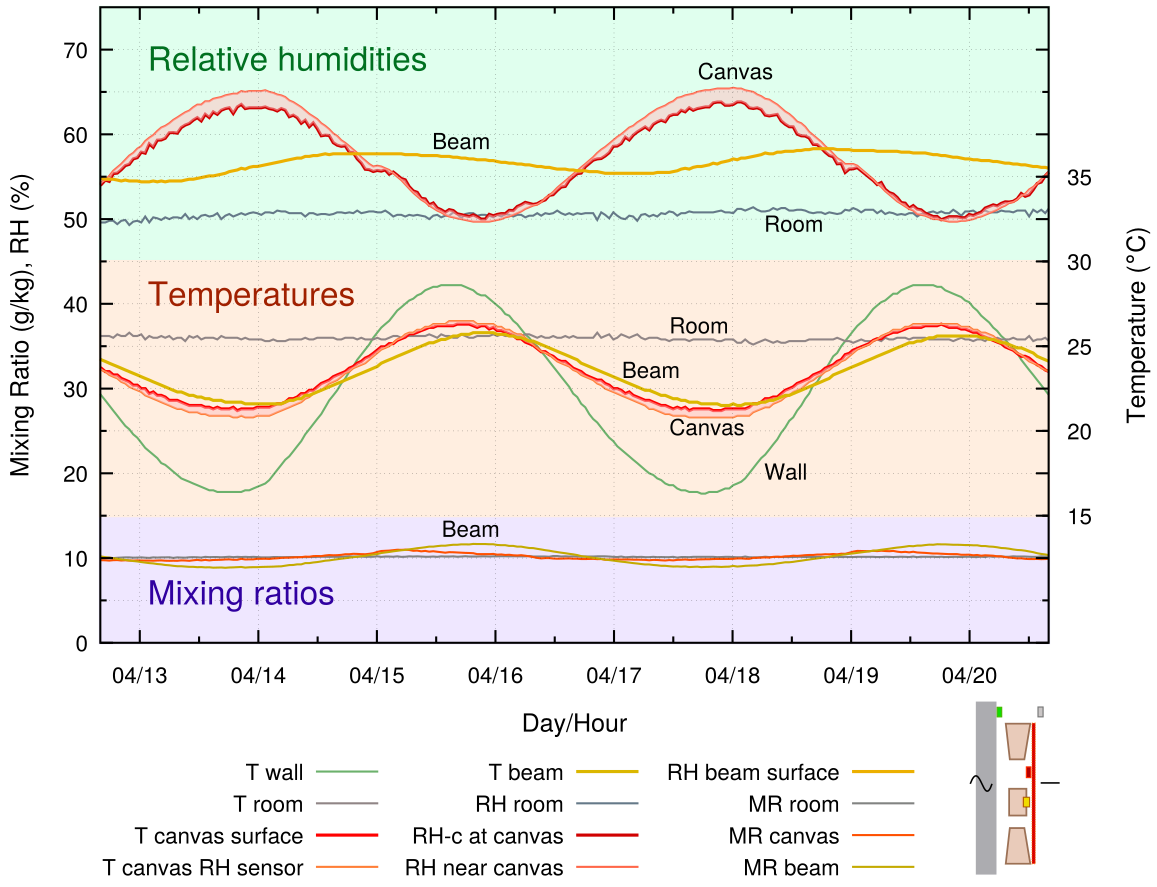
Figure 4. A 4 hour cycle of wall temperature, with an average value slightly below ambient. The painting, without back protection, is mounted with a 10 mm gap between the frame and the wall. The difference between the RH measured at the sensor 1.5 mm from the back of the canvas and the RH calculated at the nearby surface temperature is shown by a tinted band. The calculated value is named RH-c in the key. The sensor temperature and the canvas surface temperature are also linked by a tinted band.
All the graphs in this article have the same pattern, with RH curves clustered at the top, temperatures in the middle (with scale on the right side) and mixing ratios in a band at the bottom of the graph. A thumbnail of the structure is shown at the bottom right. Two RH curves are given, the measured value close to the surface of the canvas and the value calculated from the surface temperature of the canvas. The two curves are linked by a coloured fill, which is pink for the canvas and, in all the other graphs, blue for the RH close to the back plate.
This first experimental graph imitates the normal situation of a painting with an unprotected back and ample circulation of room air behind it, usually because the top of the painting is tilted away from the wall, but here caused by a uniform 10 mm gap between frame and wall.
The canvas temperature is approximately half way between ambient and the wall temperature. The vapour concentration at the canvas surface is the same as that in the room, as shown by the nearly identical and flat mixing ratio curves for the room and for the canvas. The RH varies strongly in opposite phase to the canvas temperature.
The RH between the canvas and the wooden cross beam is much more stable. The mixing ratio at this sensor deviates from the room value. This indicates vapour exchange with the wood, and with the adjacent canvas, in this poorly ventilated space.
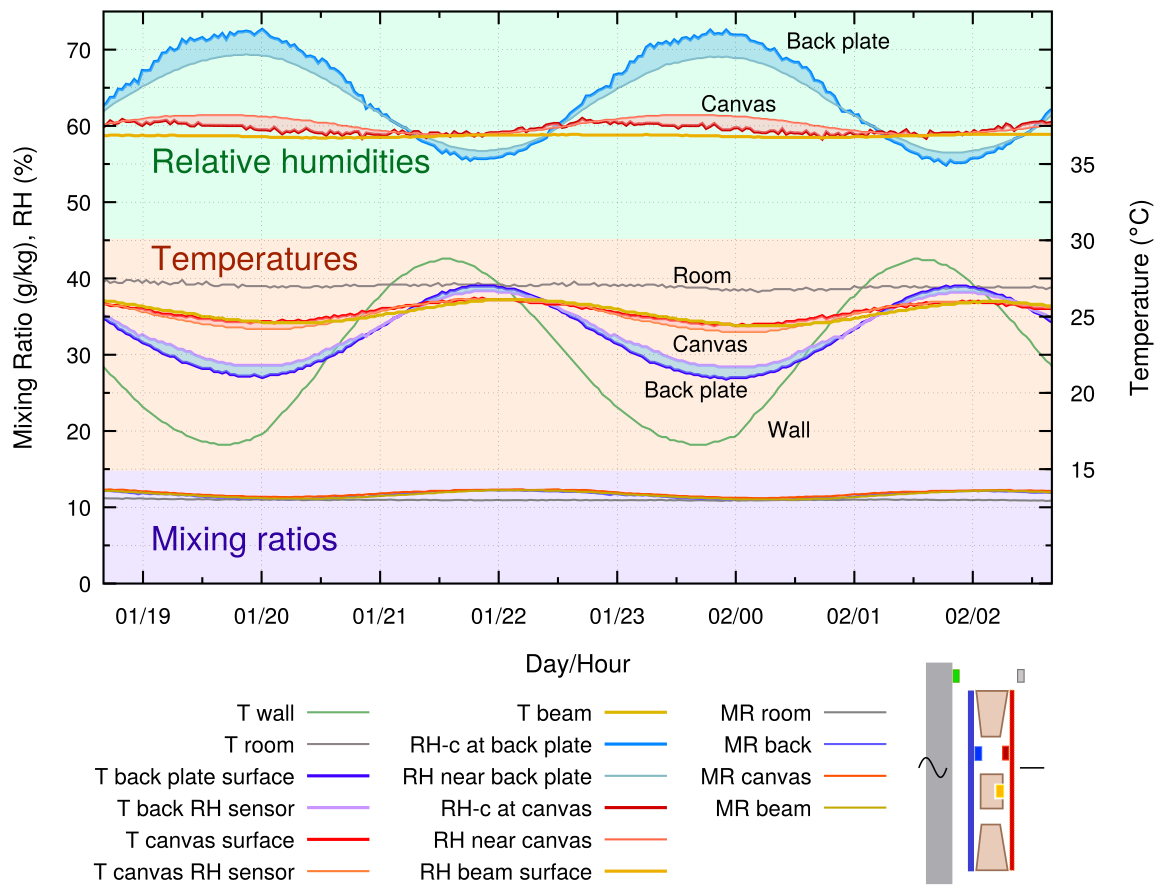
Figure 5. The painting frame is backed by an aluminium plate and held 10 mm from the wall. There are added curves for the temperature and for the RH close to the back plate.
An impermeable, moisture inert plate of aluminium is fastened to the back of the frame, but is separated from the wall by 10 mm. This gives a completely different microclimate in the sealed enclosure. There is a nearly constant RH behind the canvas and the temperature cycle is reduced. The RH stability comes from humidity buffering by the painting’s canvas, as revealed by the varying mixing ratio, indicating repeating evaporation and absorption of vapour into the confined space. The RH close to the cross beam is very stable. However, the RH at the back plate surface varies strongly over the temperature cycle.
The varying RH at the moisture-inert metal back plate can be understood by reference to two well established diagrams, the psychrometric chart (widely available on the internet) and the sorption isotherms of cellulose (Urquhart and Williams 1924).
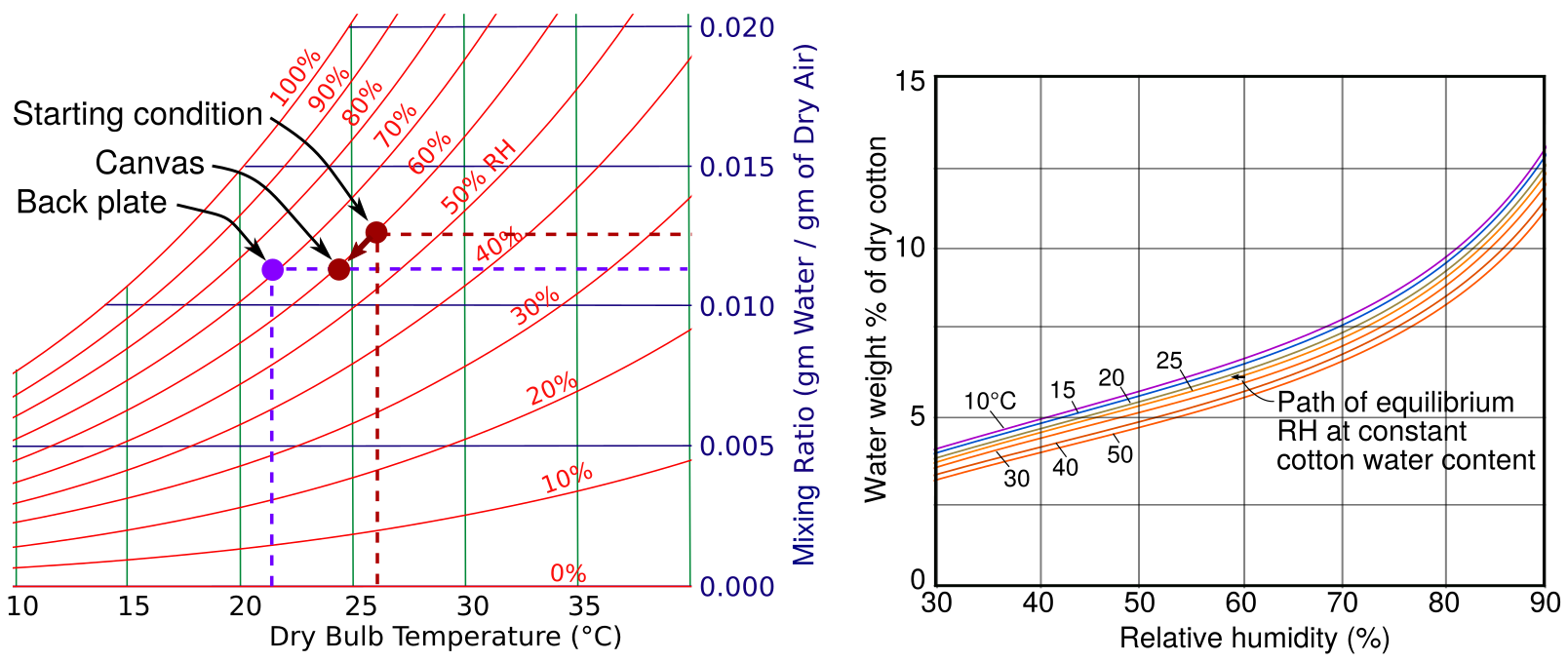
Figure 6. A simplified psychrometric chart (left) and part of the sorption curves for cotton, redrawn after (Urquhart and Williams 1924)
The starting point for the graph in figure 5 is a uniform environment throughout the assembly at 26 °C and 60% RH. When the temperature at the canvas surface declines to 24 °C at the low point of the temperature cycle, the RH at the canvas surface falls only about 1%, as defined by interpolation on the graph of sorption isotherms. So the system moves on the psychrometric chart diagonally down and to the left, following close to the curve for 60 % RH to the point marked ‘canvas’. This corresponds to a lower mixing ratio.
The mixing ratio does not respond to temperature change because a falling temperature will increase the density of the mixture but not the ratio between water molecules and air molecules. A changing mixing ratio indicates that vapour is entering or leaving the air through humidity buffering by materials. Also, its variation in space indicates the degree of mixing of the air within the enclosure. In this context, one can regard air as entraining water molecules and helping to distribute them evenly throughout the space. There is no interaction between water vapour and air other than random elastic collisions; one should not regard air as a solvent for water vapour.
As the air close to the canvas disperses out into the space behind it, the mixing ratio does not change, because there is no source or sink of water molecules to change the ratio of water molecules to air molecules. When this packet of air reaches the surface of the back plate it is at a point on the diagram which is horizontally at the same level as the ‘canvas’ point, because the mixing ratio has not changed, but it is at a lower temperature. It is at a point which intercepts the curve for 72 % RH.
The mixing ratio has no significance for the wellbeing of the art which this air touches. The potency of water vapour for chemical reaction and sorption into solid materials is expressed by the relative humidity. This switching of focus between vapour concentration, MR, as the cause of vapour movement in space, and vapour potential, RH, as a cause of change to artefacts, puzzles many people.
Further confusion has been injected by an assertion by some conservators, notably in conference session question periods, that allowing a valued artefact to exchange water vapour with its environment is unethical: an object should not be expected to control its own environmental RH. However, in a confined space the exchange of vapour between solid and air is negligible, as a quick calculation will reveal.
The volume enclosed by the painting and its back plate and frame is 0.006 cubic metres. This volume of dry air weights 7.6 grams at 25 °C. The variation of mixing ratio arising from a five degree temperature change, without significant change of RH, is about 2g/kg, which is 15 milligrams of water exchange in this enclosure.
The exposed area of canvas weighs about 180 g. So its weight change is 0.015/180 × 100 = 0.01 %, compared with an exchangeable water content in the cotton of 6.5% (at 60% RH).
Good stability of RH is achieved by very little water exchange by the canvas within the small enclosed volume.
The unprotected canvas exposed to a varying temperature and the constant mixing ratio of the room air, as in figure 4, will exchange much more water, because its surface RH will change substantially, but the mixing ratio will not change, being held constant by flushing with room air.
Evidently, a tight enclosure around the painting allows it to control its surface RH with hardly any moisture exchange. It is the surface RH which matters; how it is achieved is irrelevant. The addition of an impermeable, non-absorbent back plate is beneficial to a painting on canvas.
The large amplitude of the RH variation at the back plate means that if the wall is very cold, there is a possibility of condensation on the inside surface of the back plate. Water can run down and collect at the bottom of the frame, causing damage to the lower edge of the painting.
The 10 mm gap between painting and wall is therefore important because it reduces the temperature swing at the back plate. It also allows room air to warm the wall surface where air circulation is restricted by the proximity of the painting and thus inhibit condensation on the wall surface.
When the back plate is set directly against the wall, its RH exceeds 80% when the wall temperature reaches 16 °C. For this reason, fitting a back protection must be combined with thermal separation from the wall, by a space or by insulation.
Protection against condensation on the interior of the back plate can be provided by putting thermal insulation outside the enclosure, between the wall and the aluminium back plate.
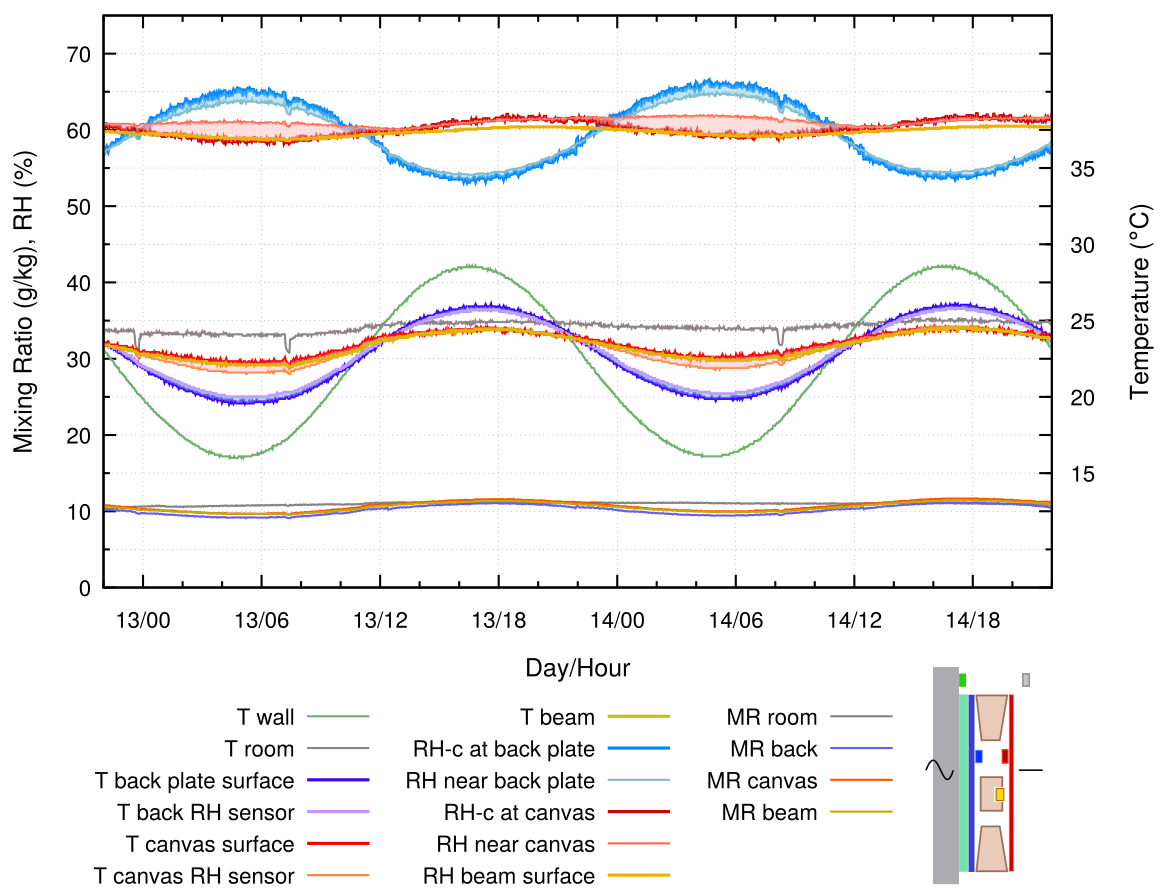
Figure 7. The empty enclosure behind the painting is separated from the wall by a 20 mm slab of expanded polystyrene. 24 hour cycle.
The RH variation with external insulation of the back protected painting is quite moderate, because the temperature variation at the back of the frame is half of that when the back plate touches the wall. However, the point of danger has now been shifted to the wall surface behind the insulation, which is now much colder than the exposed wall. This is a particular danger in humidified exhibition rooms in cool climates.
Historically, many backboards have been moisture absorbent materials such as wood and card.

Figure 8. Wooden planks screening the stone wall behind a niche which usually holds a painting. Curia Church, Floriana, Malta. Photo by Valentina Lupo.
To simulate these materials, a piece of washed, dense cotton canvas (632 g/m2) was stuck to the inside surface of the aluminium plate.
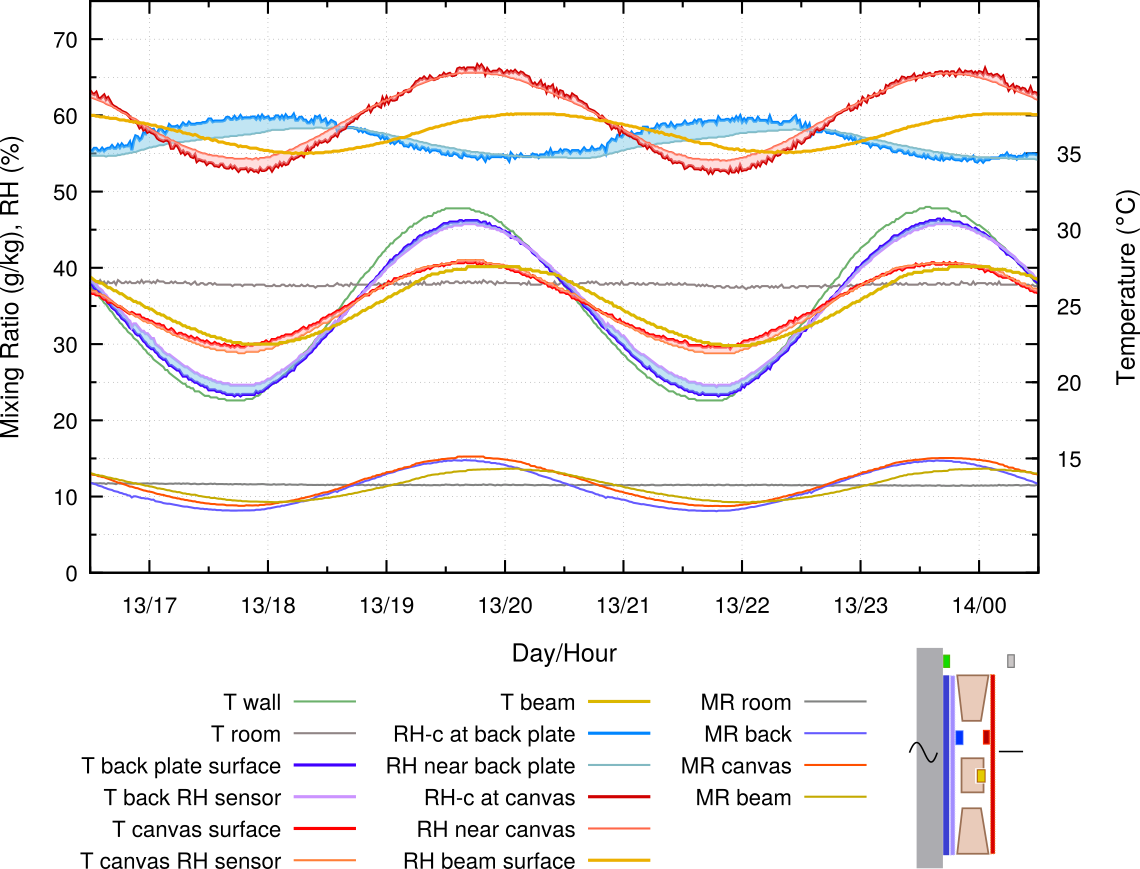
Figure 9. Buffering by canvas stuck to the inside surface of the back plate, which is set against the wall. 4 hour cycle.
The RH cycle amplitude behind the painting canvas is now rather large, and the RH cycle measured close to the cotton on the back plate is smaller than when the back plate was bare.
The process here is a more complicated version of that proposed for figure 5. In that construction the painting canvas was the main provider of water vapour. When the back plate is also hygroscopic it will compete for influence over the humidity within the enclosure.
As the temperature descends to a minimum, the RH at the back plate will tend to rise, but the back plate is hygroscopic and will absorb water vapour to move its surface RH towards equilibrium with its moisture content. The surface air of reduced mixing ratio will migrate to the painting canvas, which is about three degrees warmer. During the journey through the temperature gradient to a warmer surface its mixing ratio will not change, so its RH will decrease. At the painting canvas surface, vapour will be released into the unexpectedly dry air, in an attempt to keep the surface RH in equilibrium with the canvas moisture content. The canvas moisture content will therefore decrease.
The two hygroscopic surfaces are competing as humidity buffers, which is why the observed RH change at the back plate is less than it was in the absence of the buffer surface. The painting canvas RH span is however of greater amplitude than when there was no buffer at the back.
The mixing ratios at canvas and back plate are changing in the same direction but are not identical, indicating that the air mixing is not entirely effective in ensuring a homogenous atmosphere at this rapid temperature cycle. Nevertheless, the mixing ratios are moving in phase, though the RH values at the two hygroscopic surfaces are in opposite phase.
This graph is an example of an important and widespread phenomenon - the destabilisation of the RH around an artefact when its buffer ‘protector’ is transiently at a different temperature. It is a risky decision to put a water absorbent back plate on a painting.
This phenomenon has been neglected in the centuries-long debate over back protection of paintings. Concern over leakage into the painting enclosure of a hostile room RH has dominated the discussion. Humidity buffers play two distinctly separate roles: they serve to maintain a constant RH during temperature change, as shown by the temperature insensitivity of the cellulose isotherms, and they replace vapour leaking from the enclosure into the different mixing ratio in the room, even when there is no temperature gradient at all. This is because of the huge quantity of exchangeable water in the buffer compared with the tiny amount of water in the small air space. There is abundant literature describing how to buffer against leakage, with calculations to predict how much buffer is needed to give a desired time delay for the move towards equilibrium with the room climate.
If water vapour exchange with the room is an important consideration, due to a persistently unsuitable environment, one can add extra humidity buffer, so long as it is always at the same temperature as the painting.
Putting an extra humidity buffer close to the canvas, and thus at a similar temperature, will not cause competition between the absorbent surfaces and it will slow down the RH change caused by leakage. For this to succeed, there must be thermal insulation to shield the buffer from the back plate temperature.
The same canvas that was used as the back buffer was instead fastened to the front of a 20 mm thick slab of expanded polystyrene insulation which fitted between the back plate and the painting canvas. It was spaced 3 mm behind the painting canvas to avoid spoiling the taut contours of the painting. The twenty four hour cycle is shown in figure 10.
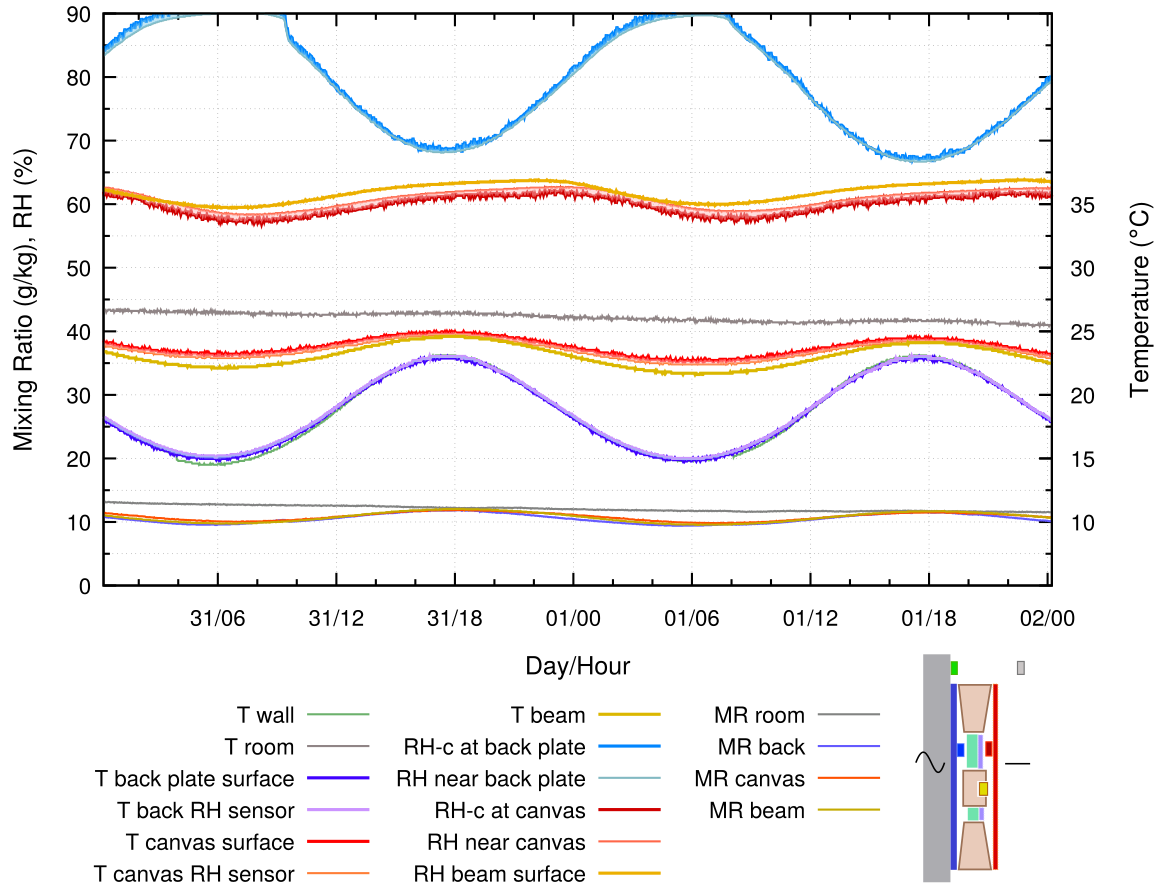
Figure 10. Enclosure with a buffer layer of cotton fixed over 20 mm of insulation and spaced 3 mm from the canvas, measured over a 24 hour cycle and at a low wall temperature.
The RH stability of the painting canvas is now very good, as is the RH between canvas and cross beam.
This combination of sealed back, impermeable insulation and a buffer layer close in temperature to the painting canvas, gives stability to both temperature and relative humidity at the back surface of the painting.
The disadvantage of this arrangement is a large temperature difference between buffer and back plate and a correspondingly high risk of condensation on the inside surface of the back plate.
The mixing ratio in the narrow unbuffered void between the insulation and the back plate is varying in the same way as in the buffered space behind the painting canvas. This shows good air mixing throughout the space, even with reasonably close fitting insulation panels within the frame.
In winter the wall may always be cooler than the room temperature and sometimes below the dew point of the air just behind the painting. Water will condense onto the back plate from vapour provided by the buffer at the warmer room temperature. From there it can dribble down to collect at the bottom rim of the assembly. Insulation is not a universal good, just as buffer materials have to be positioned with understanding of the microclimatic implications of their presence.
This construction is best reserved for paintings with a relatively benign wall temperature but a seasonally variable room RH, where extra buffering will defend against RH change caused by air exchange through the painting, or around its frame.
The leakage rate of the picture assembly was 0.4 air changes per hour. Yet there was no discernible movement of the average inside mixing ratio towards that in the room, even over several daily cycles. Evidently, the painting canvas alone provides considerable RH buffering against leakage as well as against temperature variation.
It is difficult to give an exact buffer capacity for the picture canvas, and impossible to do this for a historic painting without a stressful measurement. We can however estimate the buffering effect of the buffer canvas which we used, which was pure cotton. A real painting would have a diminished capacity, according to the degree of impregnation with diffusion reducing materials [gregers-hoegh2019].
We first calculate the buffer value (B-value), as explained by Padfield and Jensen [padfield2011]. The concept is simple. One calculates the volume of space which is equivalent in its vapour reaction to the moisture content change in the canvas when the RH is changed. Assume a change in RH from 60% to 50%. From the data in figure 6 one finds that this change will cause the cotton to lose 0.85 weight percent. For the area of cotton buffer in our experiment this corresponds to a weight loss of one gram.
We now calculate the volume of air which will lose exactly this amount of water vapour in moving from 60% to 50% RH. This is 0.43 m3. This air volume is equivalent to the canvas in its moisture reaction.
This volume can be imagined as an enclosed volume of room air which is joined to the painting enclosure through a vapour permeable membrane, so it becomes a virtual volume added to the actual painting enclosure volume, which is only 0.0056 m3.
The painting enclosure was measured to leak at 0.4 air changes per hour. This leakage rate, converted into m3 per hour, is 0.0056 x 0.4 = 0.00224 m3.
This leakage rate by volume per hour is now applied to the 0.43 m3 of the virtual volume, which gives a time for one air exchange at 192 hours, which is 8 days.
A painting whose own buffer capacity is of this approximate magnitude will cruise over daily climatic cycles with ease, without any added buffer needed.
One remaining question is the rate of response of the canvas to a changing RH at its surface. It is about 10 minutes half time to equilibrium, based on the data used by [gregers-hoegh2019]. This is so fast compared with the temperature cycles in these experiments that it can be assumed that the entire canvas thickness is always in equilibrium with its surface RH, so the analogy with a semi-permeable membrane with zero moisture resistance is reasonable. One can imagine the measured leakage, in cubic metres per hour to be entering the virtual volume of 0.43 m3 without any delay, causing it to move asymptotically towards the room RH
One of the surprises from these experiments is the evidence for rapid equilibration of the vapour content of air throughout the space, even with close fitting slabs of insulation and a rapid temperature cycle.
Condensation behind paintings has often been observed on the wall behind paintings without back boards. The painting provides enough thermal insulation to keep the wall behind it cooler than the wall directly exposed to the room air.
Conservators, aware of this risk of condensation on the cool inner surface of the back board, have tried to avert it by seemingly intuitive solutions, such as cutting the corners of the back board, or by drilling holes through it. This won’t help if the room air has a dew point above the back plate temperature. The situation becomes quite dangerous if one takes Sandner’s advice (Sandner, Schramn, and Schaft 1993), to add Artsorb behind the painting. Condensation on the back plate will dribble down inside the frame and may come into contact with the Artsorb. This material contains encapsulated lithium chloride which escapes when it is wetted (Sá, Carlyle, and Cardoso 2015). Lithium chloride is deliquescent at 13% RH, so can only be removed from contaminated surfaces by copious washing with water. Artsorb should never be used where there is a risk of condensation water reaching it.
A noteworthy phenomenon is the ‘over-buffering’ of the painting canvas. As the temperature rises in a sealed space enclosing abundant cotton, the RH rises with increasing temperature, at the rate of about one percent per three degrees rise in temperature. This can be calculated from the original data of (Urquhart and Williams 1924). In this figure the RH rises 4% for a temperature rise of less than three degrees. The most probable explanation is that there is air circulation from the cold back plate and from the considerable area of cold wood surface at the back, which equals 40% of the painting’s canvas area. This process gives a muted version of the destabilising effect seen in the back buffered experiment, figure 9. The 24 hour cycle gives time for the relatively slow acting wood surfaces to play their hygroscopic role. However, sealing the back of the frame with polyethylene tape did not remove this over-buffering phenomenon.
Nowadays many paintings have glass in front. This extra insulation layer will result in a greater temperature variation at the painting canvas as the wall temperature varies, but a smaller temperature difference between front and back of the enclosure. This results in a smaller RH cycle amplitude at the back plate and thus a diminished risk of condensation. Figure 11 can be compared with the unglazed equivalent, figure 10.

Figure 11. Enclosure with a buffer layer of cotton fixed over 20 mm of insulation and spaced 3 mm behind the painting canvas. This is the same construction and wall temperature cycle as in figure 10 but with added glass, 3 mm thick, spaced 6 mm in front of the painting. The temperature cycle amplitude at the canvas is increased, and its average value decreased, which reduces the RH variation at the back plate.
The glass prevents leakage of air through the paint surface, so this construction is well suited to paintings which hang in a room with unsuitable RH.
In massive buildings the wall may have a more stable temperature at its inner surface than the air in the room. This situation is mimicked in figure 12. The painting was exposed, without internal buffer and without glass, to a varying room temperature while set against a constant temperature at its back plate.
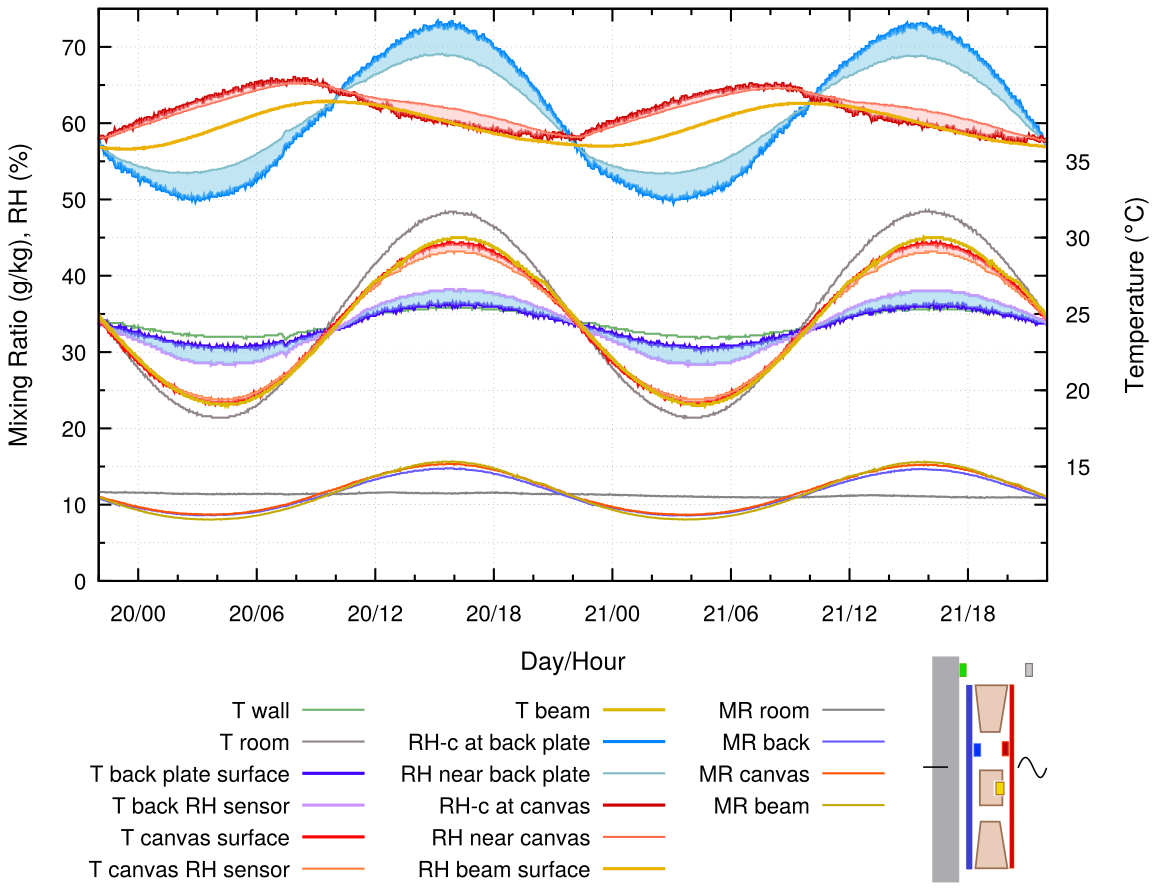
Figure 12. The empty enclosure, exposed to a varying room temperature and a constant wall temperature. 24 hour cycle.
There are several puzzling aspects of this diagram. The canvas and the beam RH are nearly in phase with each other but displaced from the temperature and the back plate RH phase. Yet the mixing ratio is uniform throughout the space and in phase with the temperature.
As the canvas temperature falls, it absorbs water vapour, as shown by the falling MR, but not enough to stem the rise in RH at its surface caused by the temperature reduction, which is now larger than when the wall was varying in temperature. As the fall in temperature diminishes and then reverses, the canvas continues to absorb vapour, because its water content is out of equilibrium with its surface RH. After a half cycle, the temperature rises above ambient and the canvas quickly begins to desorb vapour, which pushes its equilibrium surface RH down during the whole of the above ambient part of the temperature cycle. The result is a non-sinusoidal RH cycle about a quarter cycle in advance of the temperature cycle and the MR cycle.
Adding glass in front of the painting gives a similar pattern but with reduced amplitude of the temperature cycle.
Our next experiment re-instated the internal insulation and buffer layer close to the canvas.
Figure 13 shows the relatively unsteady RH at the canvas when it has the same back protection of buffer and insulation as in figure 11 but is exposed to a variable room temperature and a constant wall temperature.
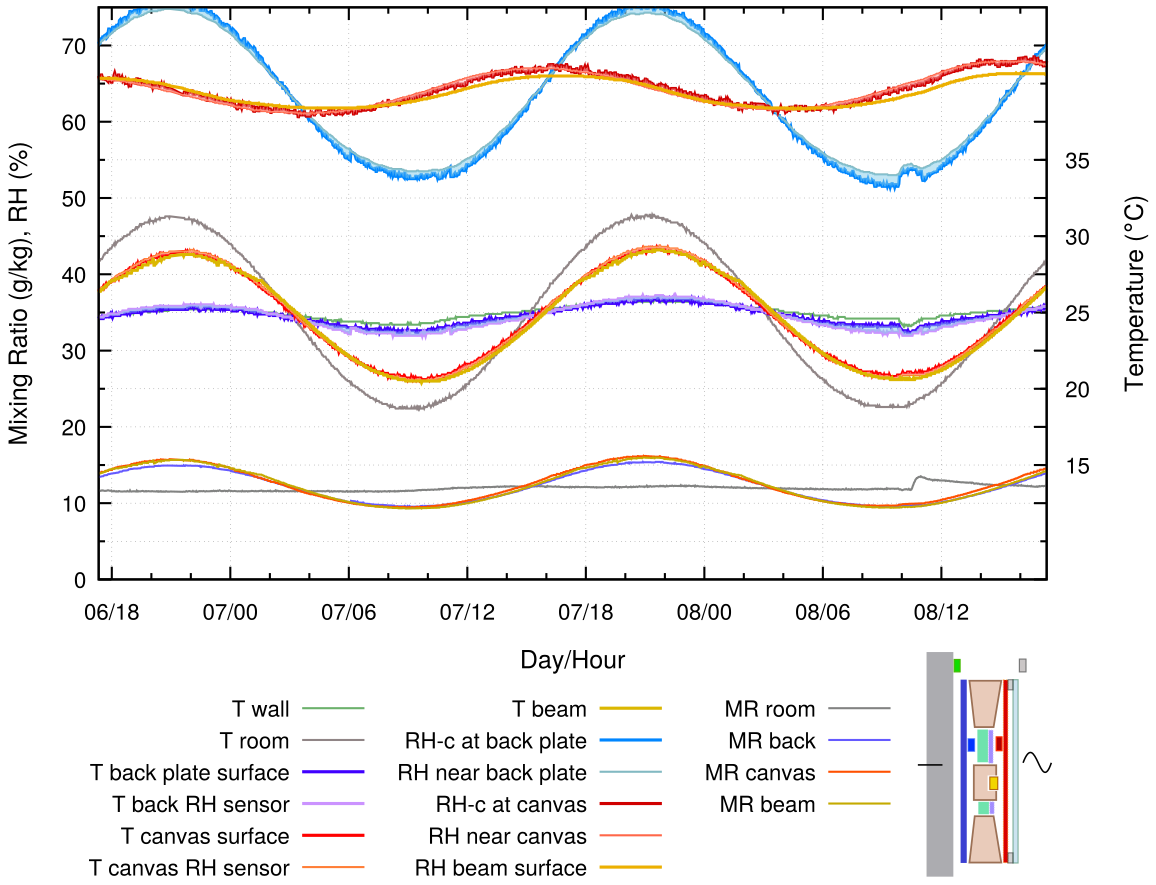
Figure 13. Enclosure with a buffer layer of cotton fixed over 20 mm of insulation and spaced 3 mm from the canvas, which is covered by glass at 6 mm separation. The glass is exposed to a temperature variation with the back plate held at a nearly constant temperature.
In this construction, the moisture reaction of the combined canvas layers dominates the entire space, so the RH at the back is variable, even though its temperature is nearly constant. The painting canvas RH is calmer but displaced by a quarter of a cycle.
Many observers have commented on the better state of preservation of oil paintings on canvas in areas which are backed, at a millimetre or two spacing, by the wooden frame on which the canvas is stretched.

Figure 14. An anonymous painting from around 1690, illuminated by raking light to show the protective effect of the wooden frame behind the canvas. 390 x 460 mm. From the Rosenborg Palace collection, Copenhagen. Photo by Berit Møller.
All the graphs show a reduced RH cycle amplitude in the 3 mm space between the wooden cross beam and the canvas. The cross beam is mounted horizontally, so convecting air currents will flush this space. Nevertheless, the 24 hour cycles show a significant calming of the RH in this region.
The temperature difference between the beam and the canvas over open space is quite small, even in the empty enclosure set directly against the wall. (Ligterink and Di Pietro 2007) attributed the stretcher effect to the thermal insulation afforded by the wood, which reduces the RH fluctuation through moderating the temperature, without a significant contribution from sorption processes. They applied the temperature gradient as a single step change, followed by a steady state temperature difference.
Our cyclic experiments are closer to the real situation and reveal, in figure 4, that for the unprotected painting, the temperature is not much different between the beam-protected area and the exposed canvas, while the RH variation at the beam surface is much less than at the exposed canvas. The mixing ratio reveals moisture exchange with wood and canvas. This combination of buffering by both wood and canvas, together with constriction of air movement in the narrow space, as shown by the diverging mixing ratio curves, seems an entirely adequate explanation for the better preservation of canvas paintings over the frame beneath.
We strove for airtightness to simplify interpretation of the experiments. However, we did not actually achieve better than 0.4 air changes per hour, even though our canvas was painted with fresh acrylic over the commercial ground. Air exchange will tend to force the mixing ratio behind the painting towards that in the room. Nevertheless, we noticed no tendency for the mixing ratio within the enclosure to drift towards ambient. In a buffered space with a large reservoir of sorbed water in materials, this process can take a long time compared with the air exchange rate as measured by trace gases that do not interact with the materials.
In the insulated and buffered assembly there is a rather small volume of air compared with the perimeter seal length, so the air exchange rate is quite large. However, the small air volume has a small vapour content, so the buffer materials, the canvas and the wood, can cope with many air changes before there is a noticeable movement in the average mixing ratio of this volume towards that of the enclosing room.
On the positive side, the relatively rapid, yet safe, air exchange rate prevents the accumulation of damaging vapours emitted by the components of the enclosure: painted canvas, wood and insulation. If this remains a concern to the conservator, an active carbon loaded cloth can be sneaked into the space. If the painting is likely to be against a wall which is normally cooler than the room, the carbon absorber is best placed close to the back plate, because cool carbon absorbs better. The support must be non water absorbent in this position.
The microclimate of the exhibition room should be taken into consideration. A wall which is frequently so cold that its surface temperature is sometimes below the dew point of the inside air requires special consideration. A painting hanging against such a cold wall, or even spaced away from the wall, will inhibit the wall warming from contact with the room air, without entirely suppressing penetration of room air to the cold wall. There is then a risk of condensation behind the painting.
Some outer walls provide a more stable climate behind the painting than the room provides to the painted surface (T. Padfield and Robinson 1996). The site of the painting should be studied before making an elaborate construction behind the painting. It may be that glazing the front of the painting will be a more effective preservative than a backing plate.
Another relevant consideration, which is not treated here, is the effect of sudden heating by sunlight, or by spot lights, which can cause moisture movement from the painting canvas to the interior of the void behind it. This was described by (T. Padfield et al. 2002).
Light energy absorbed by different colours will cause horizontal movement of water vapour behind the painting. A one degree difference in temperature corresponds to a 3% difference in RH in the space immediately behind the painting at that point.
Figure 15 summarises the performance of the enclosure when it is exposed to a cyclic temperature gradient. It shows the span of the RH and temperature changes for each enclosure structure, normalised to the wall temperature span.
The merit of each construction is judged by how small a RH span arises at the back surface of the painting canvas. In several constructions, there is a large span in RH close to the back plate. This can cause damage within the assembly, but not directly to the painting. A small temperature span at the painting is also good, but temperature is not as strong a cause of dimensional change as the RH span, under the same environmental cycle.
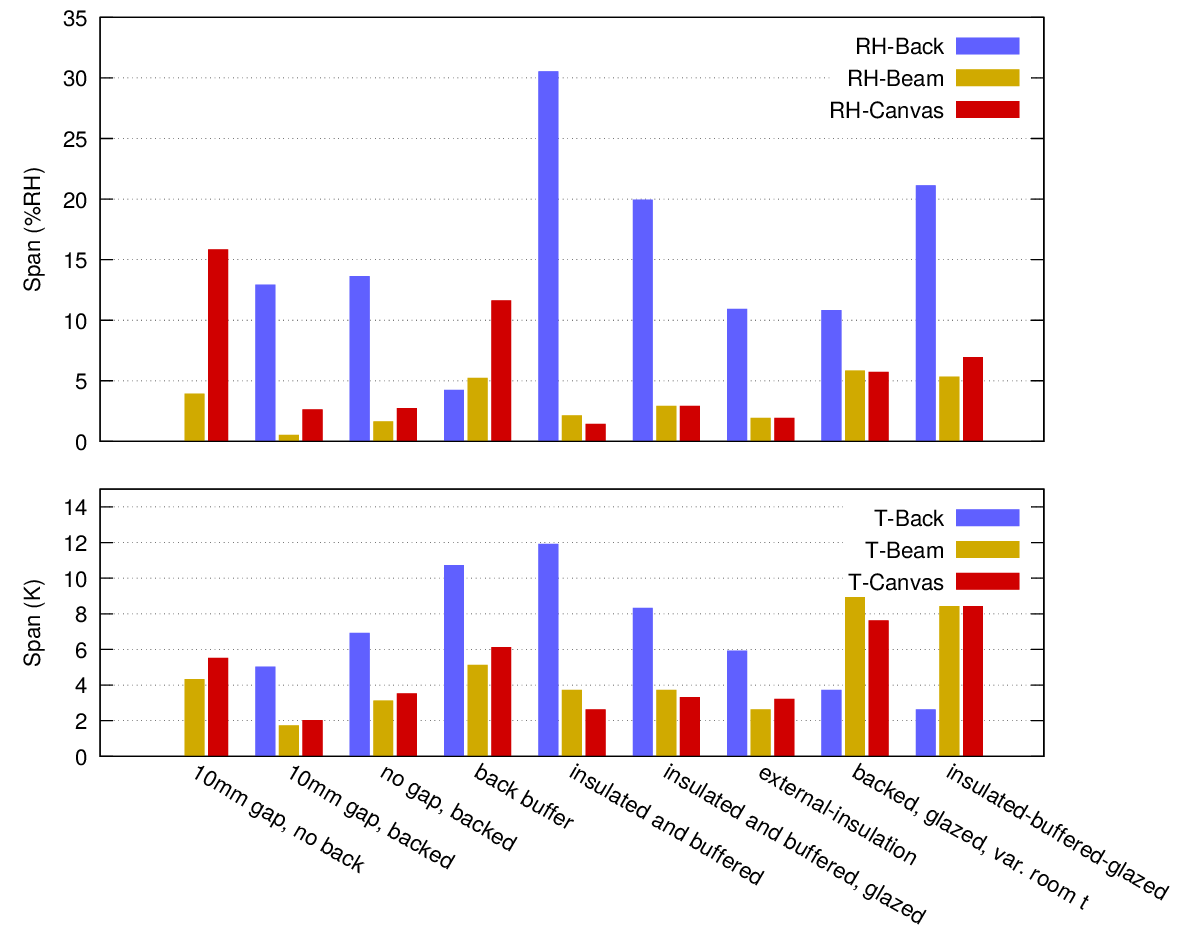
Figure 15. A synoptic view of the variability of the temperature and RH at three places in the painting enclosure. The RH variation is the measured variation at sensors adjacent to the backboard, near the painting canvas and at the painting side of the wooden beam. The rightmost two sets of columns represent a steady wall temperature with varying room temperature.
The diagram shows clearly the main features of the various treatments. The unprotected painting showed the largest variation of RH, entirely caused by its temperature divergence from the room temperature.
Backing the frame with a moisture inert and impermeable metal sheet greatly stabilised the canvas RH but caused a large RH fluctuation at the surface of the back plate. This could cause condensation if the wall becomes sufficiently cold.
Insulating between the wall and the back plate gives good stability to the painting canvas and much reduces the risk of condensation on the back plate, while transferring the condensation risk to the wall behind the insulation.
Adding buffer capacity to the back plate, by using wood for example, will reduce the condensation risk there but will increase the RH variation at the painting canvas.
Bringing the buffer close to the painting canvas, with insulation behind it, will stabilise the painting canvas RH better against leakage, but will expose the back plate to a very high RH fluctuation with correspondingly high condensation risk.
Adding glass in front of the painting slightly increases the temperature span at the painting canvas but also gives a reduced span at the back plate and reduces the RH span there.
The two last clusters on figure 15 show the microclimate when the room temperature is more variable than the wall temperature. The temperature and RH spans are increased for the painting canvas and the wooden beam but the back plate has a moderate RH span, increased when there is internal insulation and buffering.
Our conclusion is that the best general back protection for a painting of moderate size and undefined position in the world is an impermeable, non-hygroscopic plate, with 10 mm spacers to separate it from the wall. This is the second cluster from the left in figure 15. This plate can be metal or it can be a transparent plastic such as polycarbonate, if the conservator wants to be able to see the back of the canvas. Metal has the advantage of reducing radiative heat transfer; its total impermeability is not a significant advantage in this situation.
Hygroscopic back boards are dangerous because even a small fluctuation in temperature difference between back board and painting canvas will cause a large RH swing at the canvas. The Canadian Conservation Institute (Hartin 2016) recommends hygroscopic backs. This advice is good for situations where the temperature is uniform but the room RH is seasonally low, or high. It is therefore important to consider the geographical setting of the painting, as well as the room conditions in front of it.
The current authoritative advice on back protection of paintings fails to warn about the high RH which can arise at the back plate when there is a transient low temperature arising from the outside weather.
There is no reason to ventilate the space behind the painting. The space is unlikely to have an air exchange rate lower than once per day, because of the large perimeter seal compared with the enclosed volume. The often expressed fear of mould growth in the sealed space is justified, because of the high RH at the back board when it is cooler than the painting. However, ventilation with room air through perforations will not help, because it introduces air, often humidified in museum galleries, of a moisture content which may well have a dew point close to the backboard temperature. If condensation on the back plate is judged likely, a remedy can be borrowed from the air conditioning industry: separate the bottom of the back plate from the frame by a thin fabric whose threads wick the descending water down past the frame to evaporate on the outside of the assembly.
A subtle effect which shows in our experiments is over buffering, meaning that the painting canvas experiences a rise in RH beyond the small rise expected from a uniform temperature increase in a buffered space. This is caused by water vapour released from the wood surfaces facing the back plate. The wood surfaces in our experiment covered 40% of the entire canvas surface area, so their influence, particularly in the daily cycle, is considerable. This phenomenon can be reduced by sealing the back of the frame timbers with a varnish, or with aluminium foil. This action is more important in the situation where the wall temperature is steady but the room temperature is variable.
There is a view, implied in the literature cited in the introduction, that humidity buffers are always beneficial, but this is wrong wherever there is a temperature difference between the protective buffer and the object in its care. The climatic situation must be analysed. Only in modern purpose-built museums is temperature uniformity likely. In historic houses and churches, the microclimate must be measured and studied to confirm the safety of adding humidity buffers behind a painting. Many outbreaks of mould growth on art can be directly attributed to a low temperature at their surface, combined with a moderate RH in the warmer adjacent air.
We have concentrated our attention on moderating RH change directly behind the painting canvas, but temperature variation is also important to the stability of the painting. Fortunately, the constructions which perform best to moderate RH also perform well on temperature stability.
These experiments were performed at the School of Conservation of the Royal Danish Academy of Fine Arts. The school also paid for the building of the apparatus. This was constructed at the Fablab at Roskilde University, Denmark. Henning Matthiesen helped with the air exchange measurement. Useful comments were sent by Thomas Biney, David Bugeja, Dario Camuffo, Sarah Staniforth, Frank Ligterink, Giovanna Di Pietro, Jørgen Wadum, David Thickett and Stephen Hackney.
Hartin, Debra Daly. 2016. “Backing Boards for Paintings on Canvas – Canadian Conservation Institute (Cci) Notes 10/10.” https://www.canada.ca/en/conservation-institute/services/conservation-preservation-publications/canadian-conservation-institute-notes/backing-boards-paintings.html.
Ligterink, Frank J., and Giovanna Di Pietro. 2007. “Canvas Paintings on Cold Walls : Relative Humidity Differences Near the Stretcher.” In Museum Microclimates, edited by T. Padfield and K. Borchersen. National Museum of Denmark. www.conservationphysics.org/mm/ligterink/ligterink.png.
Michalski, Stefan. 2005. “Risk Analysis of Backing Boards for Paintings: Damp Climates Vs Cold Climates.” In. https://www.academia.edu/856646/2004._Risk_analysis_of_backing_boards_for_paintings_damp_climates_vs_cold_climates .
Padfield, Tim, and Nicolas Padfield. 2019. “A Workshop Cool Plate.” www.conservationphysics.org/coolplate/coolplate01.html.
Padfield, Tim, and Annabel Robinson. 1996. “The Climate Behind Pictures Mounted Against the Outer Walls of the Chapel of Ledreborg, Denmark.” In Preprints of the Icom (International Council of Museums) Committee for Conservation, Edinburgh, 1:72–75. www.conservationphysics.org/ldrbrg/ldrbrg.png.
Padfield, Tim, Henriette Berg, Nina Dahlstrøm, and Anna-Grethe Rischel. 2002. “How to Protect Glazed Pictures from Climatic Insult.” In Proceedings of the Rio de Janiero Conference of the International Council of Museums - Committee for Conservation. Ed. Roy Vontobel, London: James & James (Science Publishers), 1:80–85. www.conservationphysics.org/framepic/glazedpics.png.
Portsteffen, Hans. 2016. “Stretching Canvas Paintings - Stretcher-Systems - and the Cologne Approach.” In Tensionamento E Telai, edited by Lorenzo Marchet. Il Prato.
Sá, Sara, Leslie Carlyle, and Isabel Pombo Cardoso. 2015. “Artsorb ® in Microclimate Frames: Oddy Testing to Evaluate the Corrosive Potential of Lithium Chloride and the Efficacy of Tyvek ® to Mitigate Its Effects.” https://www.researchgate.net/publication/323943498_ArtsorbR_in_microclimate_frames_Oddy_testing_to_evaluate_the_corrosive_potential_of_lithium_chloride_and_the_efficacy_of_TyvekR_to_mitigate_its_effects.
Sandner, I., H. P. Schramn, and A. Schaft. 1993. “Reverse Side Protection of Paintings - Test Result and Suggestions for a Model.” In ICOM-Cc Triennial Meeting, Washington Dc, 157–63.
Sozzani, Laurent. 1997. “An Economical Design for a Microclimate Vitrine for Paintings Using the Picture Frame as the Primary Housing.” J. American Institute for Conservation, 95–1007. http://cool.conservation-us.org/jaic/articles/jaic36-02-001.html.
Urquhart, A. R., and A. M. Williams. 1924. “Absorption Isotherm of Cotton.” J. Textile Inst., 559–72.
Cecilia Gregers-Høegh, Martin N. Mortensen, Marie E. Christensen, Cecil K. Andersen. 'Distorted oil paintings and wax-resin impregnation – A kinetic study of moisture sorption and tension in canvas'. Journal of Cultural Heritage 40 (2019) 43–48.
Tim Padfield & Lars Aasbjerg Jensen, 'Humidity buffering of building interiors by absorbent materials'. Proceedings of the 9th Nordic Symposium on Building Physics, Tampere, Finland, 2011 pp 475 - 482.
The RH sensors are Honeywell HIH series 8120. They were calibrated over two saturated salt solutions: Sodium chloride at 76% and magnesium chloride at 33%. The tests were made at three temperatures covering the span of the experimental cycles. The claimed accuracy is ± 2 %. Three of the four were within 1% of each other throughout the tests, the fourth, used to measure the room RH, was about 2% low. The sensors have built in signal processing and cannot be adjusted. However, inconsistencies were very small compared to the RH values discussed in this article, so no corrections were applied to the graphical results.
The temperature sensors were calibrated against each other through the temperature range, since three different sensor technologies were used in close proximity: type K thermocouples, Platinum 100 ohm resistance sensors and an unidentified type in the Honeywell sensors. The Honeywell sensors proved to be seriously non linear, with kinks in the temperature response, reproducible but different in the four sensors. Only a continuous ramp in temperature revealed the deviations from a linear response. This was a surprising defect in a sensor which is used in many industrial products. We took the average of the thermocouples, and the Pt100 sensors, as the true values and adjusted the raw data to concordance among all the sensors. Even a 0.3 degree anomaly in the temperature record produces strange artefacts on the graphs, because the calculated surface RH depends on two temperature sensors being equally responsive.
The ‘wall’ is an aluminium plate, A1 size (594 x 841 mm), 5 mm thick, grooved at the back to hold a coiled copper pipe, 5 mm internal diameter. Tempered water is pumped through the pipe. The water temperature is adjusted by recirculating it through an electric heater (600 W) or a heat exchanger connected to an industrial cooler (680 W) held at 7° C. The recirculation path is controlled by magnetic valves energised by solid state relays which in turn are controlled by a Raspberry Pi micro-computer running a Python script. The plate temperature is measured by duplicate four wire platinum 100 ohm sensors. A PID controller in the script ensures smooth temperature variation. Remote control of the temperature cycle is via a web page. The apparatus and program code is open source. It is described by (T. Padfield and Padfield 2019).
The data logger is also an open source project, based on a Raspberry Pi micro-computer and scripted in Python. The sensors are generic type K thermocouples, Platinum 100 ohm resistance sensors and Honeywell HIH 8120 combined humidity and temperature sensors. The signal processing for the thermocouples is by Adafruit MAX31856 boards, and for the Pt100 sensors by Adafruit MAX31865 boards. The Honeywell sensors have built in signal processing and send a digital signal through the I2C protocol.
The Raspberry Pi uses a web server to display a control page which provides a choice of the sensors to log, the measurement interval, and download of accumulated data. This is accessible over the internet and allows remote control of the logging.
We measured the air exchange rate with a moisture independent method. The painting assembly was flushed with nitrogen to displace most of the air, then the returning oxygen was measured with an optical method based on the quenching of fluorescence of a dye in the presence of oxygen. The air exchange rate was 0.4 per hour.
The measuring instrument was a PreSens detector (PreSens Precision Sensing GmbH, Am BioPark 11, 93053 Regensburg, Germany) which sends a short pulse of blue green light through an optical fibre cable towards the sensitive dye which is held against an airtight acrylic window at the end of a brass tube sealed through the painting stretcher.
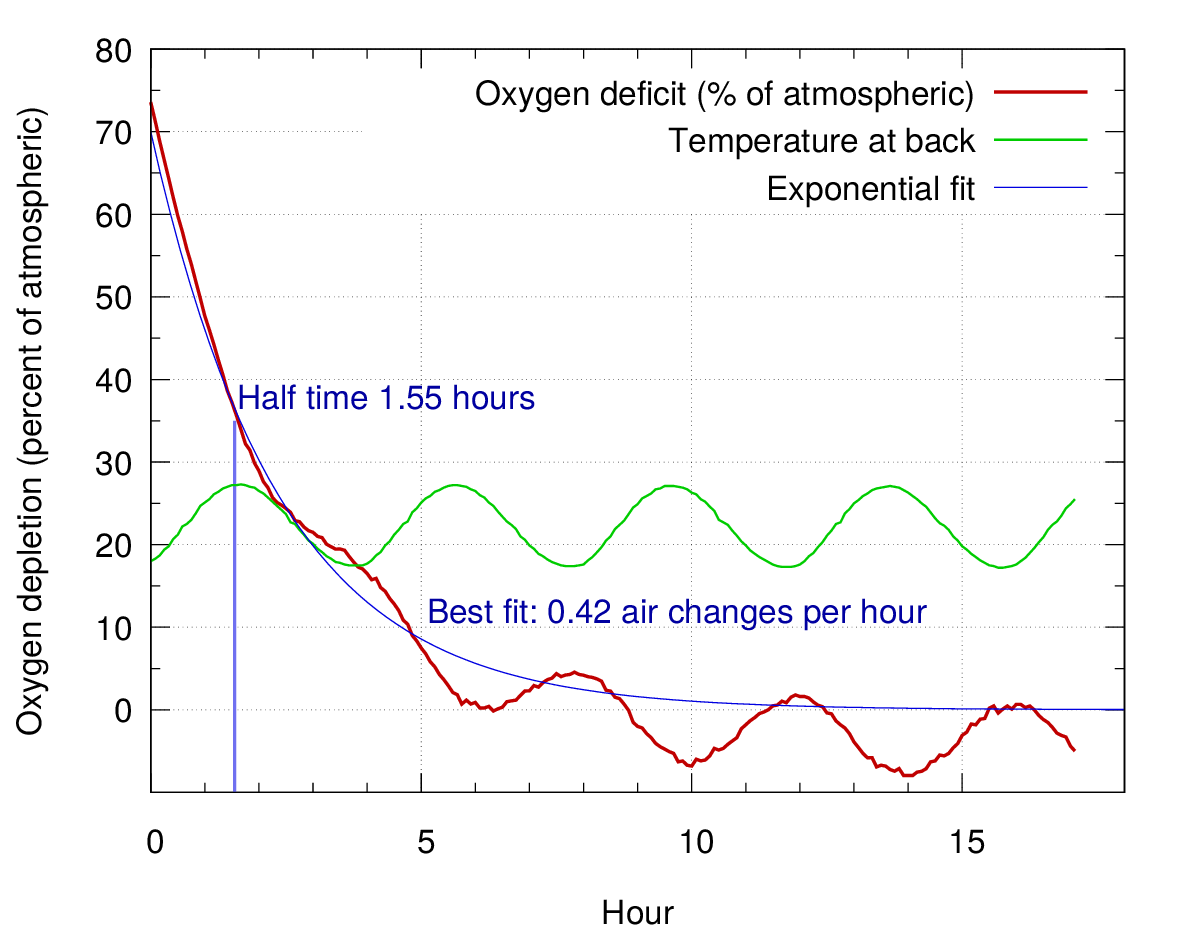
Figure 16. The air leakage rate, with a fitted curve showing the half time to equilibrium. The oscillation in the oxygen measurement is due to the large temperature dependence of the fluorescence quenching.

This work is licensed under a Creative Commons Attribution-Noncommercial-No Derivative Works 3.0 License.